Being a researcher, you will no doubt be aware of fluorescence microscopy. You may be using this tool already, or beginning work in the field, but either way, I hope you gain further insight into this field after reading this article. I’ll explain what fluorescence actually is, why certain proteins and dyes are fluorescent, where it is used and how the technique has advanced and developed.
What exactly is fluorescence?
If you type ‘definition of fluorescence’ into Google, you’ll get the following result;
“The visible or invisible radiation produced from certain substances as a result of incident radiation of a shorter wavelength such as X-rays or ultraviolet light.”
From the viewpoint of someone interested in fluorescence for microscopy, ‘radiation’ simply means light. This can include ultraviolet, infra-red and visible light. The ‘certain substances’ in the definition are referred to as ‘fluorophores’. Fluorophores have a special property whereby they can re-emit light of a higher wavelength following exposure to light of a lower wavelength.
The unit used to define and measure wavelength is the nanometre (nm). Fluorophores are defined and categorised by their excitation and emission wavelengths. The ‘shorter wavelength’ light in the definition is analogous to the ‘excitation’ wavelength. This light (or photon) is absorbed by the electrons of the fluorophore and the energy of the photon ‘excites’ the flurophore from its ‘ground’ state. When the fluorophore returns to the natural ground state, it emits a photon of a lower energy (or longer wavelength). The whole cycle of excitation/emission/ground lasts for only approximately 0.5 to 20 nanoseconds in flurophores used for microscopy.
The excitation and emission wavelengths of fluorophores are abbreviated as the lambda symbol with either a subscript ‘ex’ or ‘em’ (i.e. lex/lem).
The difference in the excitation and emission wavelengths is the property which is exploited and detected in microscopy. As individual fluorophores have distinct excitation/emission properties, then these profiles can be used to differentiate separate targets of interest in the same tissue section or cell.
Jablonski and Stokes
The first illustration of the absorption and subsequent emission of light by fluorophores was published in 1933. A Polish physicist named Professor Alexander Jablonski (1898-1980) described the cycle in a simple and effective diagram which highlights the activation of a fluorophore from its ground state to the excited state and the return to ground with the emission of a photon. Known simply as a ‘Jablonski Diagram’;
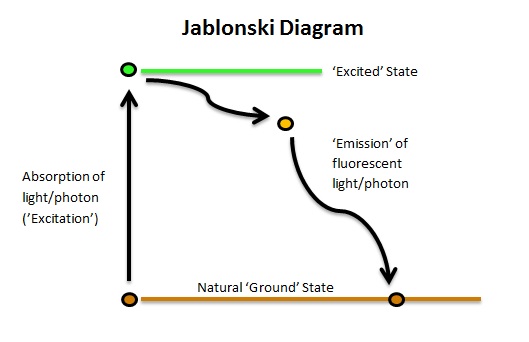
It should be noted that there can many different ground and excited states. In addition, a fluorophore may not return directly to ground, but can pass through what are known as ‘Triplet States’ of decreasing energy.
An Irish mathematician and physicist, Sir George Gabriel Stokes (1819-1903), made many discoveries and advances in the fields of light and optics. In 1852, he published a paper called ‘On the Change of Refrangibility of Light’. This is a wonderful and descriptive paper unlike the research papers we are used to reading at present;
“…a ghost-like gleam of pale blue light shot right across the tube.”
You can read the complete paper here;
http://rstl.royalsocietypublishing.org/content/142/463.full.pdf+html
The ‘Stokes Shift’ was named in his honour. This is the difference between the peaks of the excitation and emission wavelengths (as measured in nm). Every single fluorophore has a distinct Stokes Shift.
Green Fluorescent Protein
Although the history of fluorescence stretches back to 1560, it wasn’t until 1955 that Davenport and Nicol described the first fluorescent component of a living organism. They isolated and examined the ‘photogenic tissue’ from a jellyfish (Aequorea victoria). It was a further seven years until the photogenic component of the jellyfish was recognised as a protein by Shimomura of Princetown University. He isolated the protein from ‘squeezates’ which was namely the solution resulting from squeezing the bioluminescent parts of the jellyfish through a cotton bag! In 2008, he was awarded the Nobel Prize in Chemistry for his discovery of Green Fluorescent Protein (GFP) along with two American scientists (Chalfie and Tsien). In his Nobel lecture, Shimomura describes GFP thus;
“GFP was a beautiful protein but it remained useless for the next 30 years after the discovery”.
Indeed, it wasn’t until 1995 that Tsein and his group discovered a single-point mutation in GFP which not only rendered it as a useful tool for biology (making it more photostable), but also resulted in the classic excitation and emission wavelengths we know today of 488/509 nm.
At present, the GFP tools available to microscopists are not restricted to green. Thanks to the different adaptations of GFP, it is now available in colours ranging from blue to yellow. Furthermore, fluorophores are used out with the field of microscopy and are widely used to study gene expression in the form of ‘reporters’. Cells of interest can be targeted and transfected with fluorescent genetically modified plasmids or viral DNA which encode for the gene of interest. This localises to the site or organelle of interest within the cell and with fluorescent imaging, biological processes can be examined in the transfected cells.
The term ‘fluorescent probe’ is usually applied to an antibody which is tagged with a fluorophore. Previously, the two most common fluorophores used were FITC (fluorescein isothiocyanate) and TRITC (tetramethyl rhodamine isothiocyanate). With numerous advances in the field, the current range of fluorescent probes, labels and proteins is vast.
One of the most common ranges of fluorescent tools is the ‘AlexaFluor’ dyes available from Molecular Probes. Alexa Fluor dyes cover the excitation wavelength spectrum from 346 to 784 nm with an array of different kits and antibodies. Similarly, many other companies offer ranges of their own probes. For example, the ten ‘DyLight’ probes from Thermo Scientific ten cover the fluorescence spectrum from 350 nm to 800 nm.
Quantum Dots
In the 1980’s a scientist by the name of Louis Brus was working at the AT&T Bell Laboratories. He was investigating the unique properties of semiconductor particles which had very large surface areas. During these investigations he made a rather curious discovery- the fluorescent properties of these nanomaterials was related to their size. These were subsequently called ‘quantum dots’ (or ‘QDots’) and as the size of the quantum dots decrease, the wavelength of the light which they emit increases. Quantum dots are now available as a very bright and stable tool to the field of fluorescence microscopy. Quantum dots are approximately 20-times brighter than other ‘traditional’ fluorophores and are reported as being up to 100-times more stable in terms of signal, photobleaching and so on. Quantum dots can be also be conjugated to target proteins and due to their very large surface area, it is possible to have many target conjugations to a single QDot.
http://www.microscopyu.com/articles/livecellimaging/fpintro.html
http://www.scholarpedia.org/article/Fluorescent_proteins
http://www.piercenet.com/method/fluorescent-probes#types_of_fluorophores
Author: Martin Wilson


